PNU 100766
linezolid
CAS: 165800-03-3
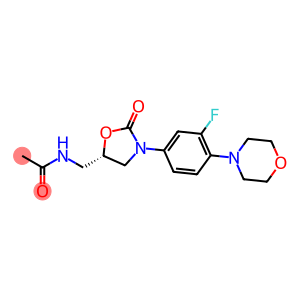
Molecular Formula: C16H20FN3O4
PNU 100766 - Names and Identifiers
PNU 100766 - Physico-chemical Properties
| Molecular Formula | C16H20FN3O4 |
| Molar Mass | 337.35 |
| Density | 1.302±0.06 g/cm3(Predicted) |
| Melting Point | 176-1780C |
| Boling Point | 585.5±50.0 °C(Predicted) |
| Specific Rotation(α) | D20 -9° (c = 0.919 in chloroform) |
| Flash Point | 307.9°C |
| Solubility | Soluble in water (<1 mg/mL at 25 °C), ethanol (8 mg/mL at 25 °C), chloroform, methano |
| Vapor Presure | 1.08E-13mmHg at 25°C |
| Appearance | White powder |
| Color | white to off-white |
| pKa | 15.53±0.46(Predicted) |
| Storage Condition | room temp |
| Refractive Index | 1.553 |
| MDL | MFCD00937825 |
| Use | Is a fully synthetic oxazolidinone antibiotic |
PNU 100766 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S36 - Wear suitable protective clothing. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| RTECS | AC2720000 |
| HS Code | 29419000 |
PNU 100766 - Reference
| Reference Show more | 1. [IF=3.631] Zhiya Yang et al."5-aminolevulinic acid-photodynamic therapy ameliorates cutaneous granuloma by killing drug-resistant Mycobacterium marinum."Photodiagn Photodyn. 2022 Mar;:102839 |
PNU 100766 - Nature
Open Data Verified Data
White Crystal, melting point 181.5~182.5 °c.
PNU 100766 - Preparation Method
Open Data Verified Data
ethyl acetate, morpholine, and diisopropylethylamine were mixed, and 3,4-difluoro nitrobenzene was added dropwise with stirring, and the reaction was completed. Extracted with ethyl acetate, washed with saturated brine, dried, evaporated to dryness and recrystallized from acetone-water to give 3-fluoro-4-morpholinyl nitrobenzene. Mix reduced iron powder, water and glacial acetic acid, reflux for a certain time, Dropwise add 3 fluorine a 4 morpholino nitrobenzene anhydrous ethanol solution, after the reaction is finished, while hot filter, evaporated dry, the resulting material was extracted with ethyl acetate, washed with brine, dried and evaporated to dryness to give 3 monofluoro-4-morpholinylaniline. It is dissolved in the mixed solution of acetone and water, adding sodium bicarbonate, Dropwise adding benzyl chloroformate, stirring, Suction filtration, solid, filtrate into ice water, Suction filtration to obtain solid. The resulting solid was recrystallized from acetone-water to obtain benzyloxycarbonyl-3-fluoro-4-morpholinylaniline.
dissolve it in anhydrous Tetrahydrofuran, under the protection of ammonia, drop add butyl lithium solution, then drop add (R)-condensation of butyl ester, after reaction, add saturated ammonium chloride solution, ethyl acetate, water. The mixture was extracted with ethyl acetate, washed with saturated brine, dried and evaporated to dryness. The obtained material was dissolved in dichloromethane and triethylamine and reacted with methanesulfonyl chloride dropwise. Extraction with dichloromethane, washing with saturated brine, drying and evaporation gave the sulfonate. The sulfonic acid ester and sodium azide were dissolved in dimethylformamide for reaction. It is extracted with acetic acid ethanol, washed with saturated brine, dried, evaporated to dryness, and substituted to give the azide derivative. The derivative and 5% palladium-carbon were reacted in absolute ethanol for a certain time, filtered and evaporated to dryness. The obtained material was dissolved in tetrahydrofuran and aqueous sodium hydroxide solution, acetic anhydride was added, and extracted with ethyl acetate, saturated brine was washed with water and dried. Evaporation, silica gel column chromatography, ethyl acetate-n-hexane recrystallization, linezolid.
PNU 100766 - Introduction
PNU 100766 - Use
Open Data Verified Data
developed by Pharmacia & Upj ohn, Inc., USA, was first launched in the United States in April 2000. Antibiotics, inhibitors of bacterial protein synthesis. Linezolid on methicillin-sensitive or resistant staphylococcus, vancomycin-sensitive or resistant intestine
Bacteria, penicillin sensitive or resistant Pneumonia Streptococcus showed good antibacterial effect, also has antibacterial activity against anaerobic bacteria. Unlike other drugs, Linezolid does not affect peptidyl transferase activity. Linezolid has a unique site and mode of action, so it is not easy to cross resistance with other antibacterial drugs that inhibit protein synthesis in the positive bacteria with essential or acquired resistance characteristics, it is also not easy to induce the development of bacterial resistance in vitro. For vancomycin-resistant Enterococcus faecalis infection, concurrent and non-concurrent skin and skin structure infection.
PNU 100766 - Reference Information
| overview | renezzoli (Linezolid Zyvox,U -100766,PNU-100766, linezolidone, linezolid, morpholidoxone, morpholidoxone) is a new type of azolidinone synthetic antibacterial drug developed and produced by Pharmacia & Upiohn Company (acquired by Pfizer), which was approved by FDA on April 18, 2000. This product is the first drug approved for the treatment of methicillin-resistant Staphylococcus aureus infection in the United States in 40 years. It was listed in my country in September 2007 and belongs to medical insurance Class B drugs. |
| Indications | Used to treat infections caused by gram-positive (G) cocci, including concurrent bacteremia; Hospital-acquired pneumonia; Community-acquired pneumonia (Pneumonia is clinically divided into hospital-acquired pneumonia and community-acquired pneumonia due to different pathogenic bacteria.) and concomitant bacteremia; complex skin and skin soft tissue infections, including diabetic foot infections complicated with osteomyelitis; non-complex skin and skin soft tissue infections; treatment of vancomycin-resistant Enterococcus faecium infection. |
| use | is a fully synthetic oxazolidinone antibiotic oxazolidinone antibacterial drug antipyretic and analgesic bulk drug inhibit bacterial mRNA translation. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Supplier List
CAS: 165800-03-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 165800-03-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 165800-03-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 165800-03-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 165800-03-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 165800-03-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 165800-03-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 165800-03-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 165800-03-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 165800-03-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 165800-03-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 165800-03-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025





